Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (34): 5469-5476.doi: 10.3969/j.issn.2095-4344.0682
Previous Articles Next Articles
Evaluation of osseointegration of plasma electrolytic oxidation coated titanium implants in vivo
He Tao1, 2, Zhang Yunhui3, Zhang Chao1, Xu Zhiguo4, Dong Yuqi1
- 1Department of Traumatic Orthopedics, Shanghai Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200127, China; 2Laboratory of Biomechanics and Experimental Orthopaedics, Clinic Großhadern, Ludwig-Maximillian University, Munich 81377, Germany; 3Department of Orthopedics, Shanghai Baoshan District Hospital of Integrated Traditional and Western Medicine, Shanghai 201900, China; 4Department of Orthopedics, Shandong Energy Zaozhuang Mining Central Hospital, Zaozhuang 277100, Shandong Province, China
-
Received:2018-09-02Online:2018-12-08Published:2018-12-08 -
Contact:Dong Yuqi, Chief surgeon, Department of Traumatic Orthopedics, Shanghai Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200127, China -
About author:He Tao, Doctorate candidate, Surgeon, Department of Traumatic Orthopedics, Shanghai Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200127, China; Laboratory of Biomechanics and Experimental Orthopaedics, Clinic Gro?hadern, Ludwig-Maximillian University, Munich 81377, Germany -
Supported by:the National Natural Science Foundation of China, No. 30700170; Shanghai Key Basic Project of Shanghai Science and Technology Commission, No. 07JC14057; Shanghai Science and Technology Commission Nano Special Project, No. 0852nm03300; the Funded Project of China Scholarship Council, No. 201606230235
CLC Number:
Cite this article
He Tao, Zhang Yunhui, Zhang Chao, Xu Zhiguo, Dong Yuqi. Evaluation of osseointegration of plasma electrolytic oxidation coated titanium implants in vivo[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(34): 5469-5476.
share this article
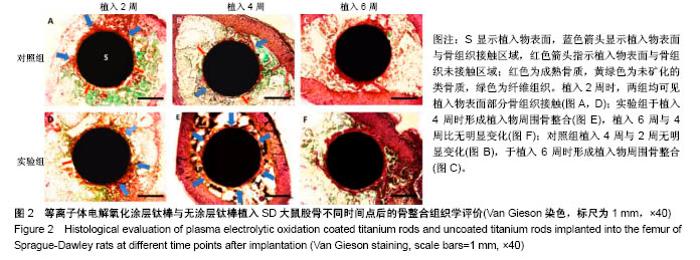
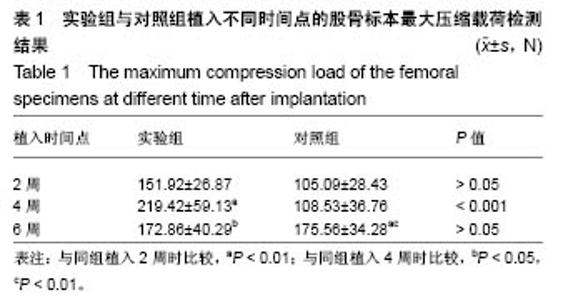
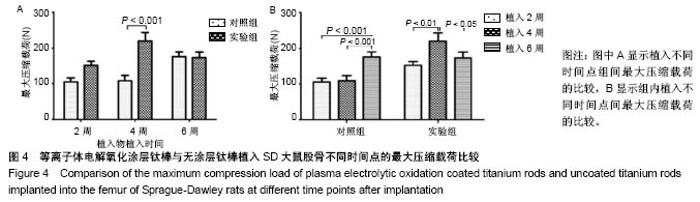
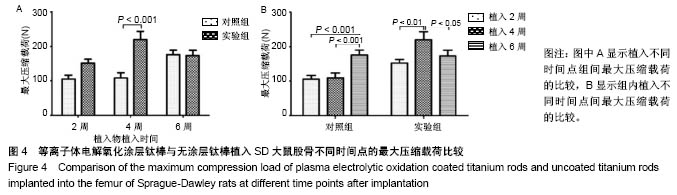
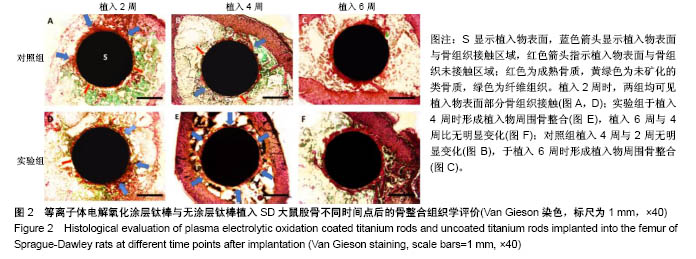
2.1 动物一般情况 36只SD大鼠术后均未出现伤口感染,体质量无明显下降,伤口愈合良好,术后2 d开始活动良好。按照实验设计时间节点,于术后2,4,6周处死,收集所有大鼠股骨标本,尸体解剖显示大鼠术后均未出现植入物相关感染。 2.2 各组标本组织学评价结果 两组在植入物-骨组织界面均未见纤维组织长入,但界面的骨组织生长情况显示出差异(图2)。植入2周(图2A,D)时,两组均显示出了植入物表面部分骨组织接触,但仍有部分表面尚未与骨组织形成紧密接触;同时两组均显示在靠近皮质骨的一侧,植入物周围的骨组织生长更加明显。植入4周时,实验组植入物表面基本被骨组织完全包绕,松质骨呈现连续性接触,植入物-骨组织界面仅可见少量空隙;对照组植入物周围骨组织接触相比植入2周时无明显区别,松质骨与植入物表面呈散在点状接触(图2B),植入物表面仍存在部分裸露区域,但未见明显纤维组织接触(图2B)。植入6周时,两组在植入物均显示出了连续的植入物-骨组织接触(图2C,F),其中实验组植入物-骨界面较植入4周时未见明显变化,尽管松质骨的厚度似乎有所增加(图2F);对照组的植入物周围松质骨骨量明显增加并与植入物形成紧密接触,同时连续性也较4周时明显增强(图2C)。"
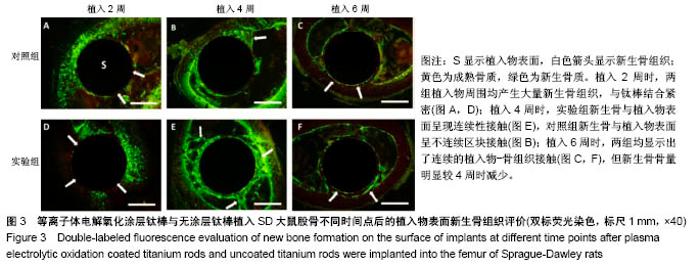
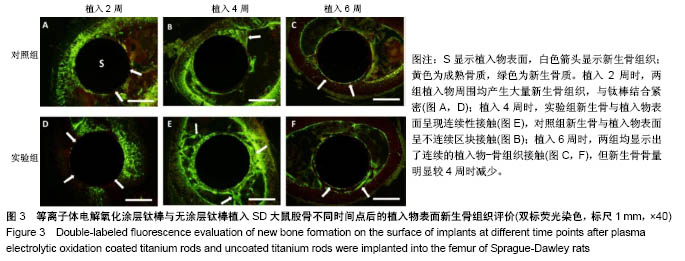
2.3 各组标本双标记荧光评价结果 双标记荧光显示,两组钛棒植入后各时间点的新生骨呈现不同的规律(图3)。植入2周时,两组均在植入物周围产生大量新生骨组织,与钛棒结合紧密,松质骨呈现密集网状结构,松质骨厚度较薄(图3A,D),新生骨呈现不连续分布,可见植入物表面存在部分裸露区域,新生骨与植入物表面呈散在点状接触(图3A,D)。植入4周时,实验组植入物表面完全被新生骨包绕,新生骨与植入物呈现连续性接触,新生骨仍然活跃而且厚度增加(图3E);对照组植入物周围新生骨组织接触面积虽有所增加,但植入物表面仍有部分区域未见新生骨接触,新生骨与植入物表面呈不连续区块接触(图3B),同时两组均显示在靠近皮质骨的一侧,植入物周围的骨组织生长更加明显(图3B,E)。植入6周时,两组在植入物均显示出了连续的植入物-骨组织接触(图3 C,F),但新生骨骨量明显较4周时减少,且仅局限于骨质的边缘及植入物-骨界面,呈线性不连续分布(图3 C,F)。"
| [1] Belatti DA, Pugely AJ, Phisitkul P, et al. Total joint arthroplasty: trends in medicare reimbursement and implant prices. J Arthroplasty. 2014;29(8):1539-1544. [2] Jager M, Jennissen HP, Dittrich F, et al. Antimicrobial and Osseointegration Properties of Nanostructured Titanium Orthopaedic Implants. Materials(Basel). 2017;10(11):E1302. [3] Perren SM, Regazzoni P, Fernandez AA. How to Choose between the Implant Materials Steel and Titanium in Orthopedic Trauma Surgery: Part 2 - Biological Aspects. Acta Chir Orthop Traumatol Cech. 2017;84(2):85-90. [4] Branemark PI. Vital microscopy of bone marrow in rabbit. Scand J Clin Lab Invest. 1959;11 Supp 38:1-82. [5] Branemark PI. Osseointegration and its experimental background. J Prosthet Dent. 1983;50(3):399-410. [6] Southam JC, Selwyn P. Structural changes around screws used in the treatment of fractured human mandibles. Br J Oral Surg. 1971;8(3):211-221. [7] Esposito M, Hirsch JM, Lekholm U, et al. Biological factors contributing to failures of osseointegrated oral implants. (I). Success criteria and epidemiology. Eur J Oral Sci. 1998; 106(1):527-551. [8] Branemark R, Branemark P, Rydevik B, et al. Osseointegration in skeletal reconstruction and rehabilitation: a review. J Rehabil Res Dev. 2001;38(2):175-182. [9] 贺韬,曹聪,董宇启.硬组织工程材料骨整合评价方法与应用[J].国际骨科学杂志,2012,33(2):92-95,117.[10] Donath K, Breuner G. A method for the study of undecalcified bones and teeth with attached soft tissues. The Sage-Schliff (sawing and grinding) technique. J Oral Pathol. 1982;11(4): 318-326. [11] 罗亚平,李莹莹,胡纯婷,等.胃泌素对大鼠激素性骨坏死的治疗作用[J].上海交通大学学报(医学版),2018,38(3):276-280.[12] He T, Cao C, Xu Z, et al. A comparison of micro-CT and histomorphometry for evaluation of osseointegration of PEO-coated titanium implants in a rat model. Sci Rep. 2017; 7(1):16270. [13] Pellegrini G, Francetti L, Barbaro B, et al. Novel surfaces and osseointegration in implant dentistry. J Investig Clin Dent. 2018:e12349. [14] Zhu X, Chen J, Scheideler L, et al. Effects of topography and composition of titanium surface oxides on osteoblast responses. Biomaterials. 2004;25(18):4087-4103. [15] Suo L, Jiang N, Wang Y, et al. The enhancement of osseointegration using a graphene oxide/chitosan/ hydroxyapatite composite coating on titanium fabricated by electrophoretic deposition. J Biomed Mater Res B Appl Biomater. 2018. doi:10.1002/jbm.b.34156. [16] Bai L, Du Z, Du J, et al. A multifaceted coating on titanium dictates osteoimmunomodulation and osteo/angio-genesis towards ameliorative osseointegration. Biomaterials. 2018; 162:154-169. [17] Huang MS, Chen LK, Ou KL, et al. Rapid Osseointegration of Titanium Implant With Innovative Nanoporous Surface Modification: Animal Model and Clinical Trial. Implant Dent. 2015;24(4):441-447. [18] Yang DH, Lee DW, Kwon YD, et al. Surface modification of titanium with hydroxyapatite-heparin-BMP-2 enhances the efficacy of bone formation and osseointegration in vitro and in vivo. J Tissue Eng Regen Med. 2015;9(9):1067-1077. [19] Dundar S, Yaman F, Bozoglan A, et al. Comparison of Osseointegration of Five Different Surfaced Titanium Implants. J Craniofac Surg. 2018. doi: 10.1097/SCS. 0000000000004572. [20] Yan JH, Wang CH, Li KW, et al. Enhancement of surface bioactivity on carbon fiber-reinforced polyether ether ketone via graphene modification. Int J Nanomedicine. 2018;13: 3425-3440. [21] Lukaszewska-Kuska M, Wirstlein P, Majchrowski R, et al. Osteoblastic cell behaviour on modified titanium surfaces. Micron. 2018;105:55-63. [22] Li NB, Sun SJ, Bai HY, et al. Preparation of well-distributed titania nanopillar arrays on Ti6Al4V surface by induction heating for enhancing osteogenic differentiation of stem cells. Nanotechnology. 2018;29(4):045101. [23] Liu P, Hao Y, Zhao Y, et al. Surface modification of titanium substrates for enhanced osteogenetic and antibacterial properties. Colloids Surf B Biointerfaces. 2017;160:110-116. [24] Zhang X, Zu H, Zhao D, et al. Ion channel functional protein kinase TRPM7 regulates Mg ions to promote the osteoinduction of human osteoblast via PI3K pathway: In vitro simulation of the bone-repairing effect of Mg-based alloy implant. Acta Biomater. 2017;63:369-382. [25] Niehaus AJ, Anderson DE, Samii VF, et al. Effects of orthopedic implants with a polycaprolactone polymer coating containing bone morphogenetic protein-2 on osseointegration in bones of sheep. Am J Vet Res. 2009;70(11):1416-1425. [26] Cunningham BW, Hu N, Zorn CM, et al. Bioactive titanium calcium phosphate coating for disc arthroplasty: analysis of 58 vertebral end plates after 6- to 12-month implantation. Spine J. 2009;9(10):836-845. [27] Wang C, Karlis GA, Anderson GI, et al. Bone growth is enhanced by novel bioceramic coatings on Ti alloy implants. J Biomed Mater Res A. 2009;90(2):419-428. [28] Bai L, Liu Y, Du Z, et al. Differential effect of hydroxyapatite nano-particle versus nano-rod decorated titanium micro-surface on osseointegration. Acta Biomater. 2018;76:344-358. [29] Mostafa D, Aboushelib M. Bioactive-hybrid-zirconia implant surface for enhancing osseointegration: an in vivo study. Int J Implant Dent. 2018;4(1):20. [30] Ghadami F, Saber-Samandari S, Rouhi G, et al. The effects of bone implants’ coating mechanical properties on osseointegration: In-vivo, in-vitro, and histological investigations. J Biomed Mater Res A. 2018. doi:10. 1002/jbm. a. 36465. [31] Jiang N, Guo Z, Sun D, et al. Promoting Osseointegration of Ti Implants through Micro/Nano-Scaled Hierarchical Ti Phosphate / Ti Oxide Hybrid Coating. ACS Nano. 2018;12(8): 7883-7891. [32] Azzawi ZGM, Hamad TI, Kadhim SA, et al. Osseointegration evaluation of laser-deposited titanium dioxide nanoparticles on commercially pure titanium dental implants. J Mater Sci Mater Med. 2018;29(7):96. [33] Krupa D, Baszkiewicz J, Zdunek J, et al. Effect of plasma electrolytic oxidation in the solutions containing Ca, P, Si, Na on the properties of titanium. J Biomed Mater Res B Appl Biomater. 2012;100(8):2156-2166. [34] Hu H, Liu X, Ding C. Preparation and cytocompatibility of Si-incorporated nanostructured TiO2 coating. Surf Coat Technol. 2010;204(20):3265-3271. [35] 胡红杰,刘宣勇,丁传贤.等离子体电解氧化制备生物活性多孔纳米TiO_2涂层[J].无机材料学报,2011,26(6):585-590.[36] Sowa M, Piotrowska M, Widziolek M, et al. Bioactivity of coatings formed on Ti-13Nb-13Zr alloy using plasma electrolytic oxidation. Mater Sci Eng C Mater Biol Appl. 2015; 49:159-173. [37] Kazek-Kesik A, Kuna K, Dec W, et al. In vitro bioactivity investigations of Ti-15Mo alloy after electrochemical surface modification. J Biomed Mater Res B Appl Biomater. 2016; 104(5):903-913. [38] Chung CJ, Su RT, Chu HJ, et al. Plasma electrolytic oxidation of titanium and improvement in osseointegration. J Biomed Mater Res B Appl Biomater. 2013;101(6):1023-1030. [39] Echeverry-Rendon M, Galvis O, Quintero Giraldo D, et al. Osseointegration improvement by plasma electrolytic oxidation of modified titanium alloys surfaces. J Mater Sci Mater Med. 2015;26(2):72. [40] Pereira BL, da Luz AR, Lepienski CM, et al. Niobium treated by Plasma Electrolytic Oxidation with calcium and phosphorus electrolytes. J Mech Behav Biomed Mater. 2018; 77:347-352. [41] He J, Feng W, Zhao BH, et al. In Vivo Effect of Titanium Implants with Porous Zinc-Containing Coatings Prepared by Plasma Electrolytic Oxidation Method on Osseointegration in Rabbits. Int J Oral Maxillofac Implants. 2018;33(2):298-310. [42] Diefenbeck M, Muckley T, Schrader C, et al. The effect of plasma chemical oxidation of titanium alloy on bone-implant contact in rats. Biomaterials. 2011;32(32):8041-8047. [43] Svalastoga E, Reimann I, Nielsen K. A method for quantitative assessment of bone formation using double labelling with tetracycline and calcein. An experimental study in the navicular bone of the horse. Nord Vet Med. 1983;35(4): 180-183. [44] Li K, Wang C, Yan J, et al. Evaluation of the osteogenesis and osseointegration of titanium alloys coated with graphene: an in vivo study. Sci Rep. 2018;8(1):1843. [45] Lee JH, Hong KS, Baek HR, et al. In vivo evaluation of CaO-SiO2-P2O5-B2O3 glass-ceramics coating on Steinman pins. Artif Organs. 2013;37(7):656-662. [46] She C, Shi GL, Xu W, et al. Effect of low-dose X-ray irradiation and Ti particles on the osseointegration of prosthetic. J Orthop Res. 2016;34(10):1688-1696. [47] Kolb A, Reinisch G, Sabeti-Aschraf M, et al. Contamination of surfaces for osseointegration of cementless total hip implants by small aluminium oxide particles: analysis of established implants by use of a new technique. J Orthop Sci. 2013;18(2): 245-249. [48] Araujo MG, Lindhe J. Peri-implant health. J Clin Periodontol. 2018;45 Suppl 20:S230-S236. [49] Klar RM, Duarte R, Dix-Peek T, et al. Calcium ions and osteoclastogenesis initiate the induction of bone formation by coral-derived macroporous constructs. J Cell Mol Med. 2013; 17(11):1444-1457. [50] Klar RM. The Induction of Bone Formation: The Translation Enigma. Front Bioeng Biotechnol. 2018;6:74. [51] Ir'yanov YM, Dyuryagina OV. Effects of a local focus of granulation tissue formed in the bone marrow cavity on reparative osteogenesis. Bull Exp Biol Med. 2014;157(1): 108-111. [52] Ribeiro C, Sencadas V, Areias AC, et al. Surface roughness dependent osteoblast and fibroblast response on poly(L-lactide) films and electrospun membranes. J Biomed Mater Res A. 2015;103(7):2260-2268. [53] Sola-Ruiz MF, Perez-Martinez C, Martin-del-Llano JJ, et al. In vitro preliminary study of osteoblast response to surface roughness of titanium discs and topical application of melatonin. Med Oral Patol Oral Cir Bucal. 2015;20(1):e88-93. [54] Deng Y, Liu X, Xu A, et al. Effect of surface roughness on osteogenesis in vitro and osseointegration in vivo of carbon fiber-reinforced polyetheretherketone-nanohydroxyapatite composite. Int J Nanomedicine. 2015;10:1425-1447. [55] Okada K, Kawao N, Tatsumi K, et al. Roles of plasminogen in the alterations in bone marrow hematopoietic stem cells during bone repair. Bone Rep. 2018;8:195-203. [56] Buenzli PR, Lerebours C, Roschger A, et al. Late stages of mineralization and their signature on the bone mineral density distribution. Connect Tissue Res. 2018;59(sup1):74-80. [57] Burr DB, Gallant MA. Bone remodelling in osteoarthritis. Nat Rev Rheumatol. 2012;8(11):665-673. [58] Ueda N, Takayama Y, Yokoyama A. Minimization of dental implant diameter and length according to bone quality determined by finite element analysis and optimized calculation. J Prosthodont Res. 2017;61(3):324-332. [59] Duyck J, Vandamme K. The effect of loading on peri-implant bone: a critical review of the literature. J Oral Rehabil. 2014; 41(10):783-794. [60] Costa ML, Achten J, Griffin J, et al. Effect of Locking Plate Fixation vs Intramedullary Nail Fixation on 6-Month Disability Among Adults With Displaced Fracture of the Distal Tibia: The UK FixDT Randomized Clinical Trial. JAMA. 2017;318(18): 1767-1776. [61] Kane P, Vopat B, Paller D, et al. A biomechanical comparison of locked and unlocked long cephalomedullary nails in a stable intertrochanteric fracture model. J Orthop Trauma. 2014;28(12):715-720. [62] Pekmezci M, McDonald E, Buckley J, et al. Retrograde intramedullary nails with distal screws locked to the nail have higher fatigue strength than locking plates in the treatment of supracondylar femoral fractures: A cadaver-based laboratory investigation. Bone Joint J. 2014;96-B(1):114-121. [63] Klein GR, Levine HB, Hartzband MA. Removal of a well-fixed trabecular metal monoblock tibial component. J Arthroplasty. 2008;23(4):619-622. |
| [1] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | Xu Yulin, Shen Shi, Zhuo Naiqiang, Yang Huilin, Yang Chao, Li Yang, Zhao Heng, Zhao Lu. Biomechanical comparison of three different plate fixation methods for acetabular posterior column fractures in standing and sitting positions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 826-830. |
| [7] | Cai Qunbin, Zou Xia, Hu Jiantao, Chen Xinmin, Zheng Liqin, Huang Peizhen, Lin Ziling, Jiang Ziwei. Relationship between tip-apex distance and stability of intertrochanteric femoral fractures with proximal femoral anti-rotation nail: a finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 831-836. |
| [8] | Song Chengjie, Chang Hengrui, Shi Mingxin, Meng Xianzhong. Research progress in biomechanical stability of lateral lumbar interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 923-928. |
| [9] | Xie Chongxin, Zhang Lei. Comparison of knee degeneration after anterior cruciate ligament reconstruction with or without remnant preservation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 735-740. |
| [10] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [11] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [12] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [13] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [14] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [15] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||